Book Appointment Now
What is Ionic Bonding? Definition, Properties & Real-Life
Meta Description:
Learn what ionic bonding is, how it forms, its properties, and real-life examples like sodium chloride. A complete beginner-friendly guide.
Focus Keywords:
ionic bonding, ionic bond definition, properties of ionic compounds, sodium chloride bond
4
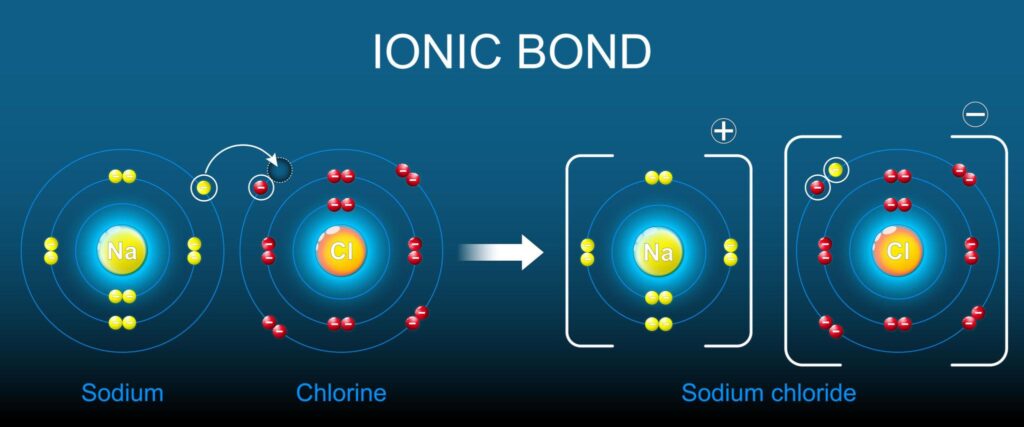
What is Ionic Bonding?
Ionic bonding is a type of chemical bonding formed through the transfer of electrons from one atom to another. It usually occurs between a metal and a non-metal.
When an atom loses electrons, it becomes a cation (+).
When an atom gains electrons, it becomes an anion (−).
Opposite charges attract — forming a strong ionic bond.
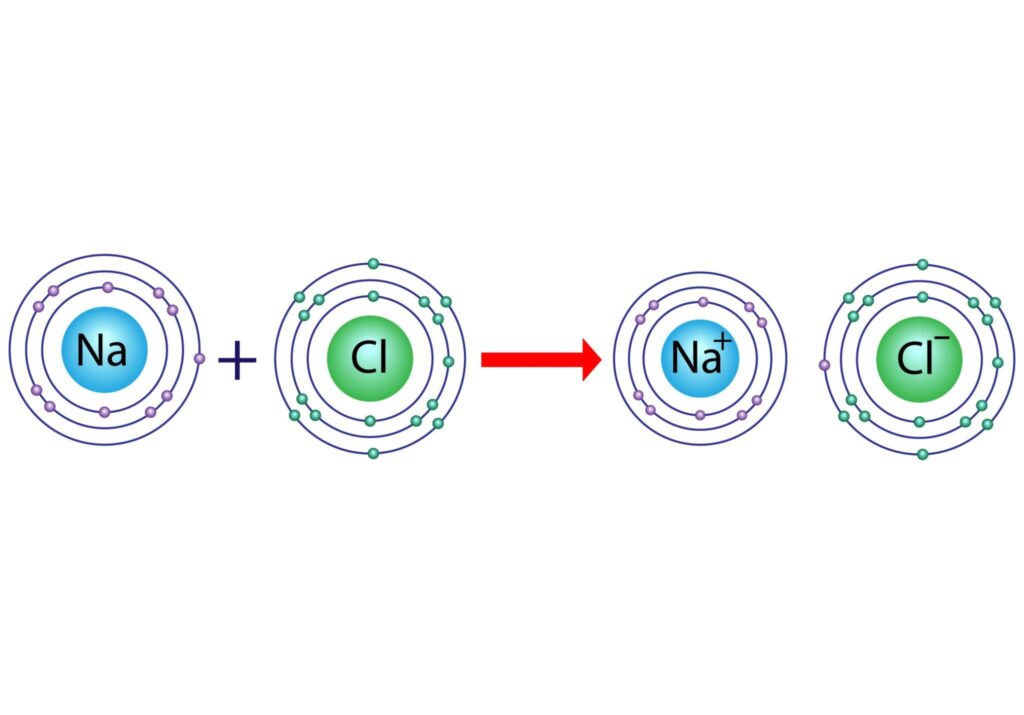
How Does Ionic Bonding Form?
Example: Sodium (Na) and Chlorine (Cl)
- Sodium loses 1 electron → Na⁺
- Chlorine gains 1 electron → Cl⁻
- Electrostatic attraction forms NaCl
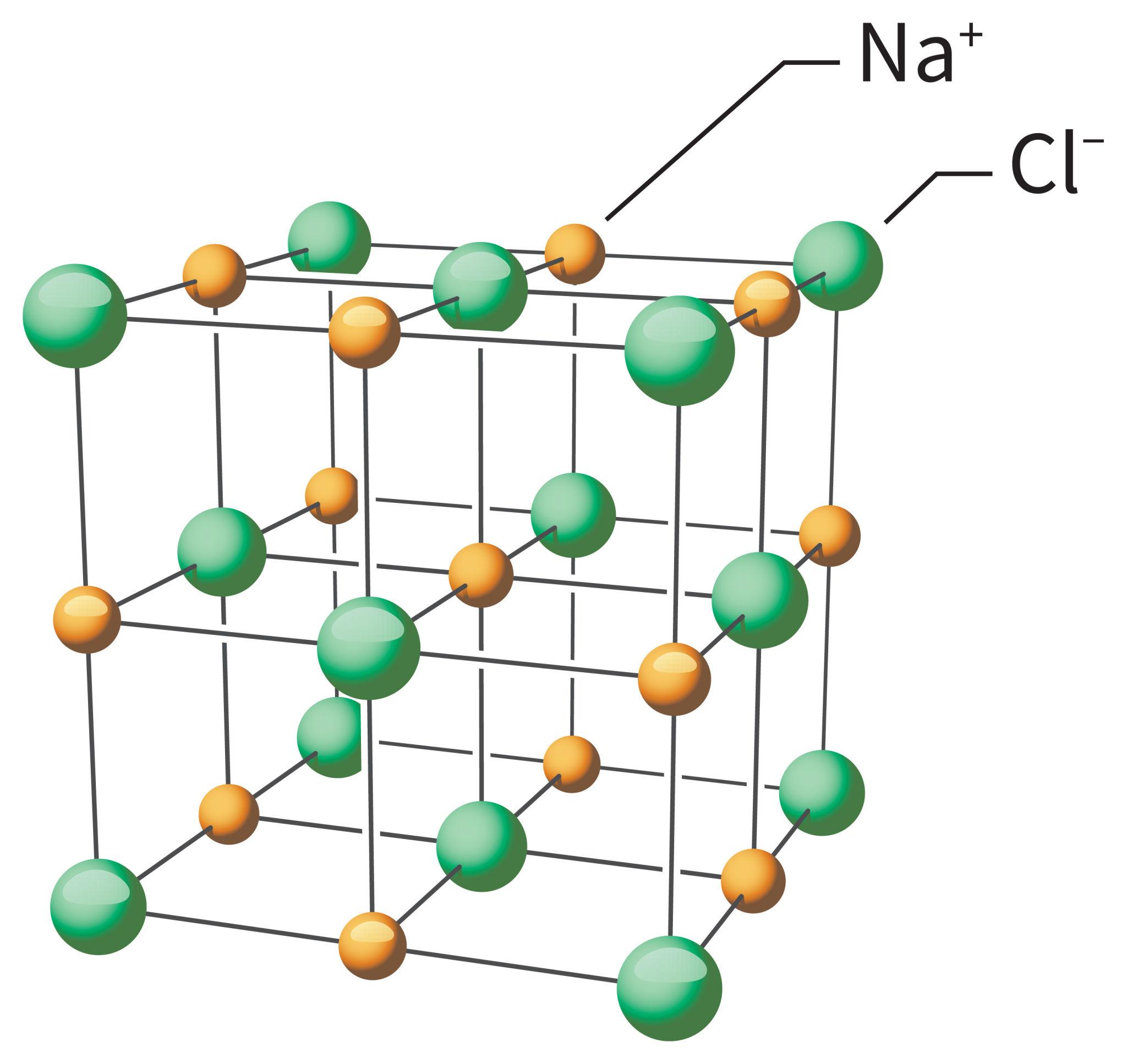
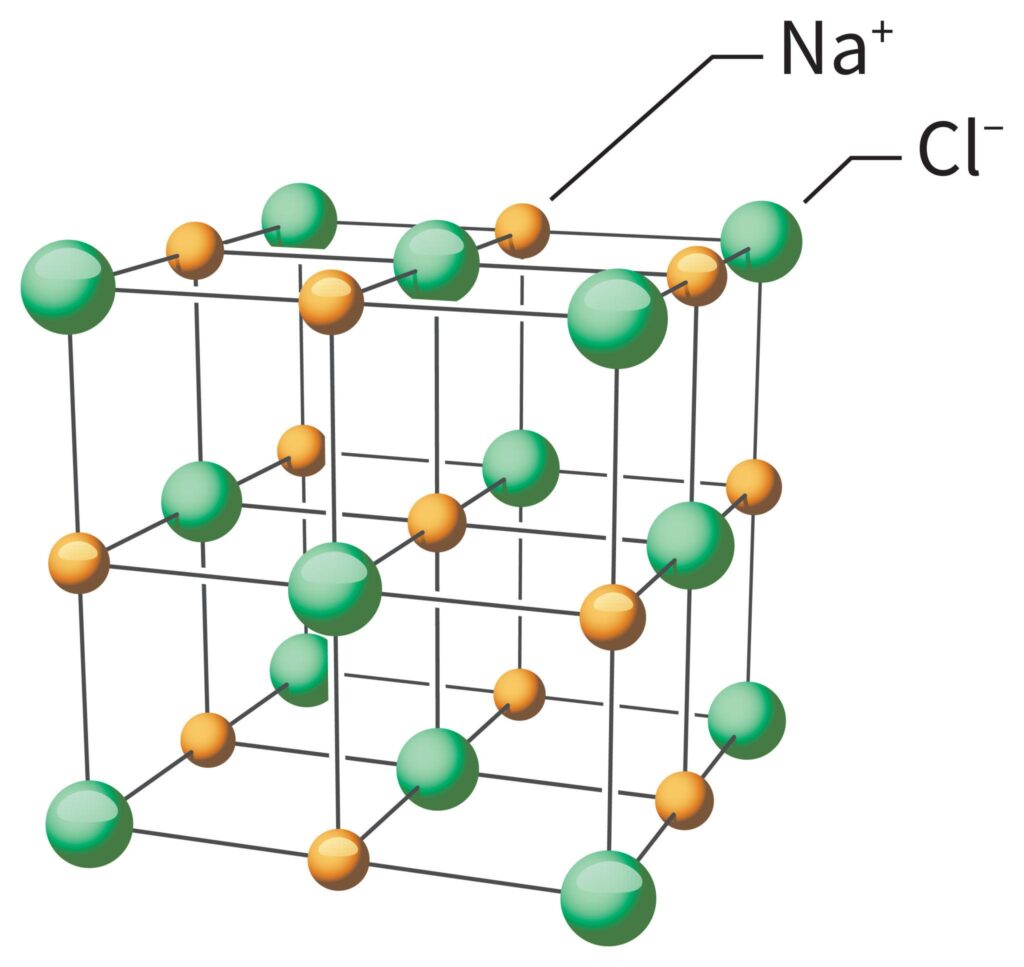
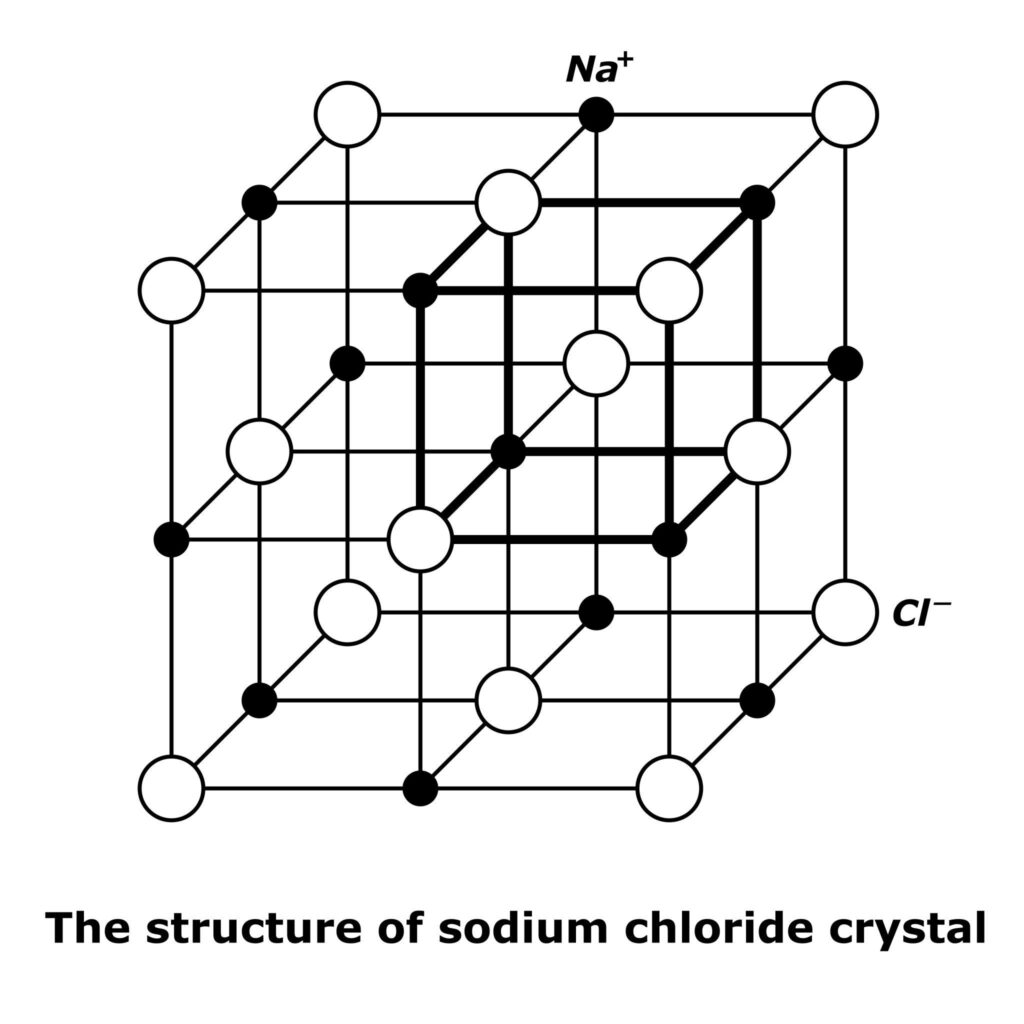
Properties of Ionic Compounds
- High melting & boiling points
- Conduct electricity when molten or dissolved
- Usually soluble in water
- Form crystal lattice structures
Real-Life Examples
- Sodium chloride (table salt)
- Magnesium oxide
- Calcium fluoride
Hashtags:
#IonicBonding #ChemistryBasics #ChemicalBonding #GCSEChemistry #ALevelChemistry #InorganicChemistry
